|
Remember how you learned the alphabet? You repeated it over and over. Repetition - How to Memorize the Periodic Table Slowly Shortcut 'Memory Hack' - How to Memorize the Periodic Table Faster than a Ferrariġ.Mental Imagery - How to Learn the Periodic Table Like a Memory Rockstar!.Songs - How to Make Repetition Less Boring.Acronyms and Acrostics - How to Memorize a Few Elements.Flashcards - How to Learn the Periodic Table Slightly Faster.Repetition - How to Memorize the Periodic Table Slowly.9.Let’s go through the top five techniques from the most common (aka the least effective and most tedious) to the Rolls Royce of memory techniques that’ll have you remembering like a memory rock star!.9.9: Periodic Trends - Atomic Size, Ionization Energy, and Metallic Character Certain properties-notably atomic radius, ionization energy, electron affinity and metallic character-can be qualitatively understood by the positions of the elements on the periodic table.9.8: The Explanatory Power of the Quantum-Mechanical Model The chemical properties of elements are determined primarily by the number and distribution of valence electrons.Electron configurations can be predicted by the position of an atom on the periodic table. 9.7: Electron Configurations and the Periodic Table The arrangement of electrons in atoms is responsible for the shape of the periodic table. 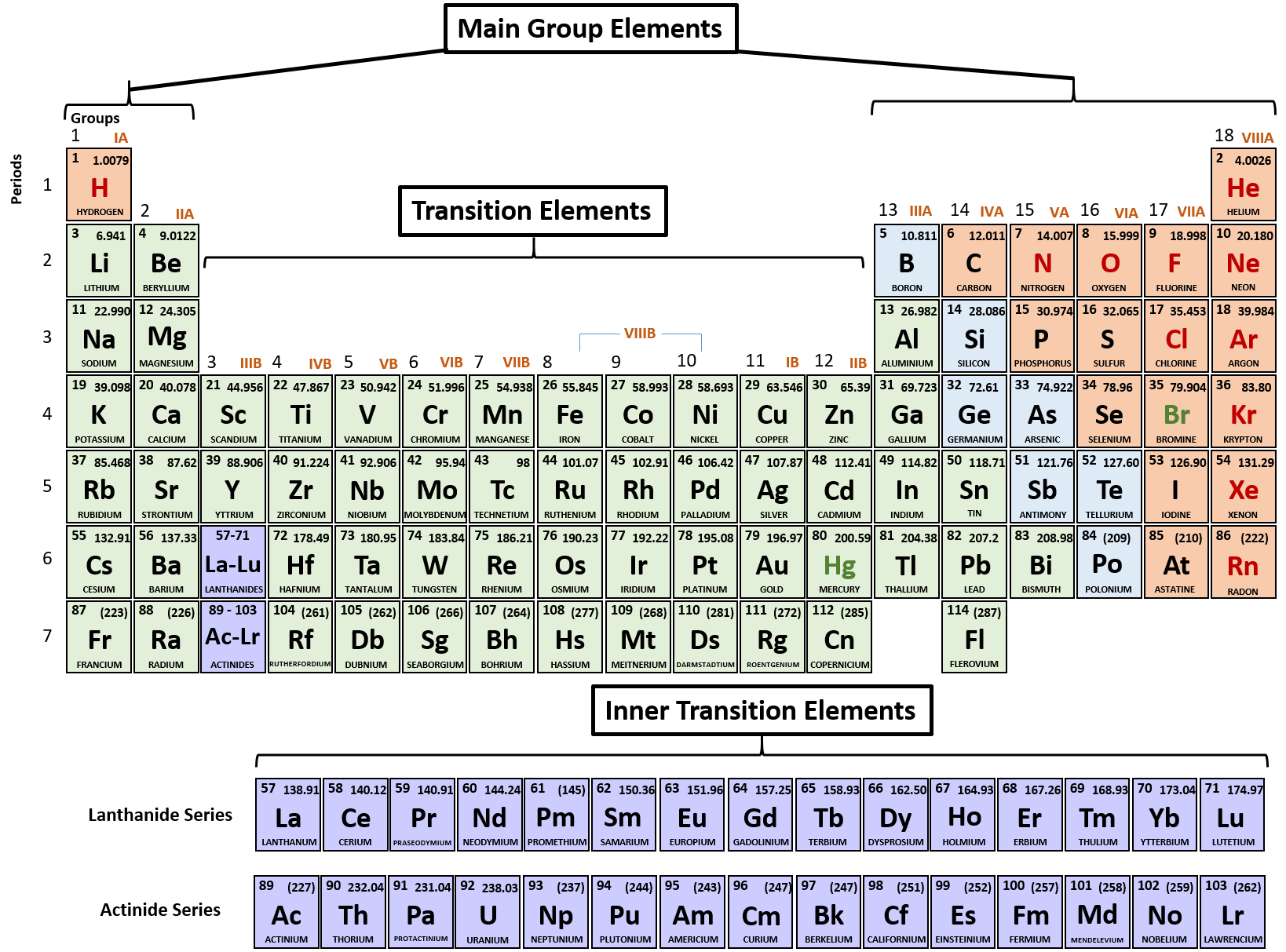
Orbital filling diagrams are a way of indicating electron locations in orbitals. Hund's rule specifies the order of electron filling within a set of orbitals. The Aufbau principle gives the order of electron filling in an atom. Electron configuration notation simplifies the indication of where electrons are located in a specific atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
